This makes gases relatively easy to compress and allows them to flow (which makes them fluids). In contrast, atoms in gases are separated by large distances, and the forces between atoms in a gas are therefore very weak, except when the atoms collide with one another. Because the atoms are closely packed, liquids, like solids, resist compression an extremely large force is necessary to change the volume of a liquid. 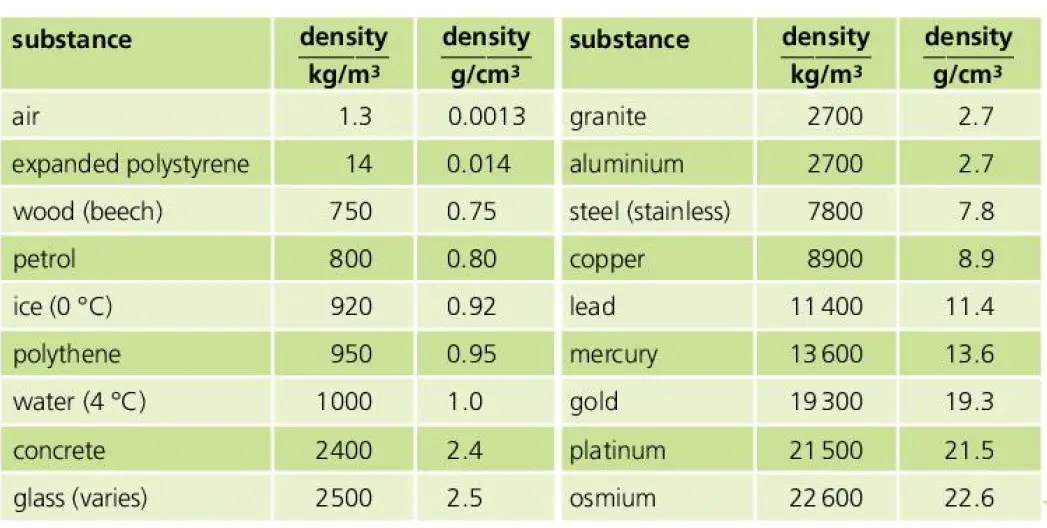
When a liquid is placed in a container with no lid, it remains in the container. That is, liquids flow (so they are a type of fluid), with the molecules held together by mutual attraction. This occurs because the atoms or molecules in a liquid are free to slide about and change neighbors. Liquids deform easily when stressed and do not spring back to their original shape once a force is removed. A gas must be held in a closed container to prevent it from expanding freely and escaping. (c) Atoms in a gas move about freely and are separated by large distances. Forces between the atoms strongly resist attempts to compress the atoms. (b) Atoms in a liquid are also in close contact but can slide over one another. If a stream had a flow of 1 ft3/s, 1 mg N03-N/L, and 0.05 mg PO4-P/L,the stream would be transporting 5.39 lbs per day of Nitrate as Nitrogen and 0.269 lbs per day of Phosphate as Phosphorus.Figure 14.2 (a) Atoms in a solid are always in close contact with neighboring atoms, held in place by forces represented here by springs. Note: mg/L x ft3/s x 2.446 = kg/day kg/day x 2.205 = lbs/dayĢ,000 lbs = one (English or long) ton - 1,000 kg = one tonne (metric or long ton) = 2,200 lbs * (14.0067 / 62.0049) = mg N/LĬalculate Loading Based on Stream Flow (cubic feet per sec or cfs) Note: Total P (mg/L as P) x 3.066 = Total mg PO₄/L Note: Total PO₄ (mg/l as PO₄) x 0.3262 = Total P Table 3 | Anion-Cation Balance: Acceptable Differenceĭealing With Common Conversions (Nutrients) and Calculating Loading Convert Total PO₄ to Total P Standard Methods for the Examination of Water and Wastewater 18th edition recommends the following guidance: The guidance on the Acceptable Percent Difference is a function of the Anion Sum. If the difference were 2 % or more, it would be very wise to conduct some additional testing. Since the difference is < 2 %, this does strongly suggest the quality of the data is good. In the example above, the percent difference between the sums of the cations and anions is positive which might suggest that a minor anion is missing. Some significant ion was not included in the testing or the lab made a mistake and reported an incorrect result (something that happens surprisingly often even in certified labs). If there is a difference, especially if it is a large difference, it means one of two things. Ideally, there should be no difference between the sum of the cations (mEq/L) and the sum of the anions (mEq/L). 
Table 2 | Cation Anion Balance - Results and CalculationsįIrst calculate the Sum of the Cations - Sum anions (SA) or 3.77397 - 3.67496 = 0.102437. This is an example of a cation-anion balance Water should be electrochemically neutral and you can use the cation and anion mass balance approach to get some idea of the potential quality of your data and attempt to understand if you are missing any specific large grouping of cations or anions. Reasons using mEq/l and expressing results in this unit is useful Reason #1: Cation and Anion Mass Balance 
If the specific gravity of the solution is not 1, the following is the relationship: A usually good approximation for fresh water is that it has a density of 1 mg/L or a specific gravity of 1. Since specific gravity is the density of a liquid divided by the density of water at 4 ☌, it has no units because they cancel out specific gravity is just a number. Specific gravity is the ratio of a liquid's density with that of water at 4 ☌, the temperature at which water is most dense, and is 999.974 kg/m3 or 0.999974 mg/L (1 mg/L = 1000 kg/m3). Density is defined as mass per unit volume, like mg/L or kg/m3 (the international standard metric unit). Note: specific gravity is related to density but they are not quite the same. For example, it is about 1.025 for sea water. The specific gravity of water is usually assumed to be ~ 1 but this can change because of temperature and amount of dissolved ions. In fresh water systems, 1 mg/L is equivalent to 1 ppm, but when the specific gravity of the solution is not 1 this is not valid. Convert from mg/L to mEq/L (milliequivalents per liter)
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
